
Smart Technology
Better Solutions
Technology in the right hands leads solutions to success.
We At SCOPE believe in technological solutions that support the way we and our customers work. Our goal is to provide flexibility based on our customer´s needs, and consequently, we select the best solutions in the market and combine them with our custom developed solutions and integrations. As A midsize Contract Research Organisation (CRO), we pride ourselves on leveraging cutting-edge technology to enhance the efficiency, accuracy, and overall quality of clinical trials. Below, you will find detailed information about our key technological solutions and how they support our mission to deliver excellence in clinical research.
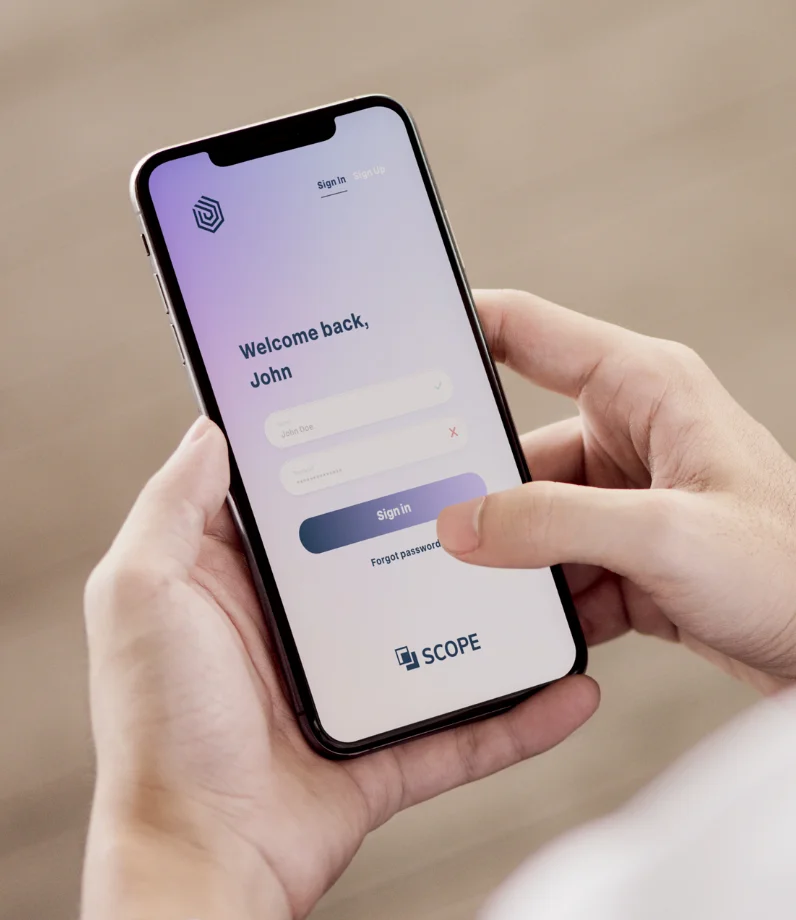
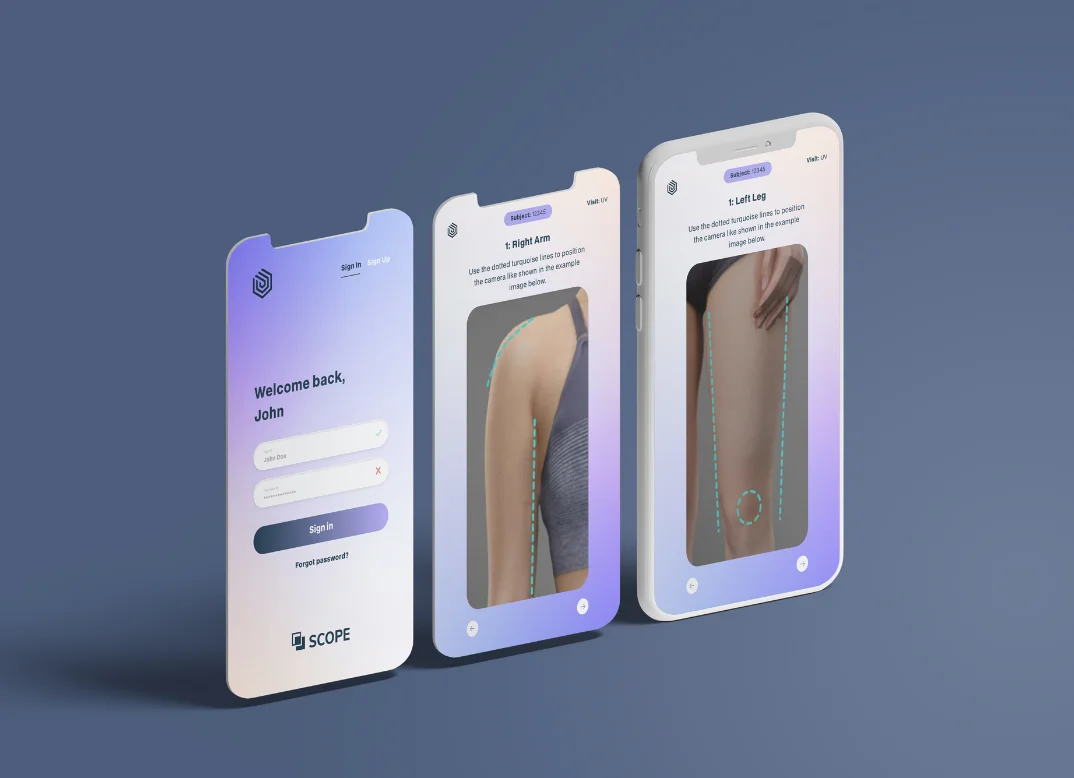
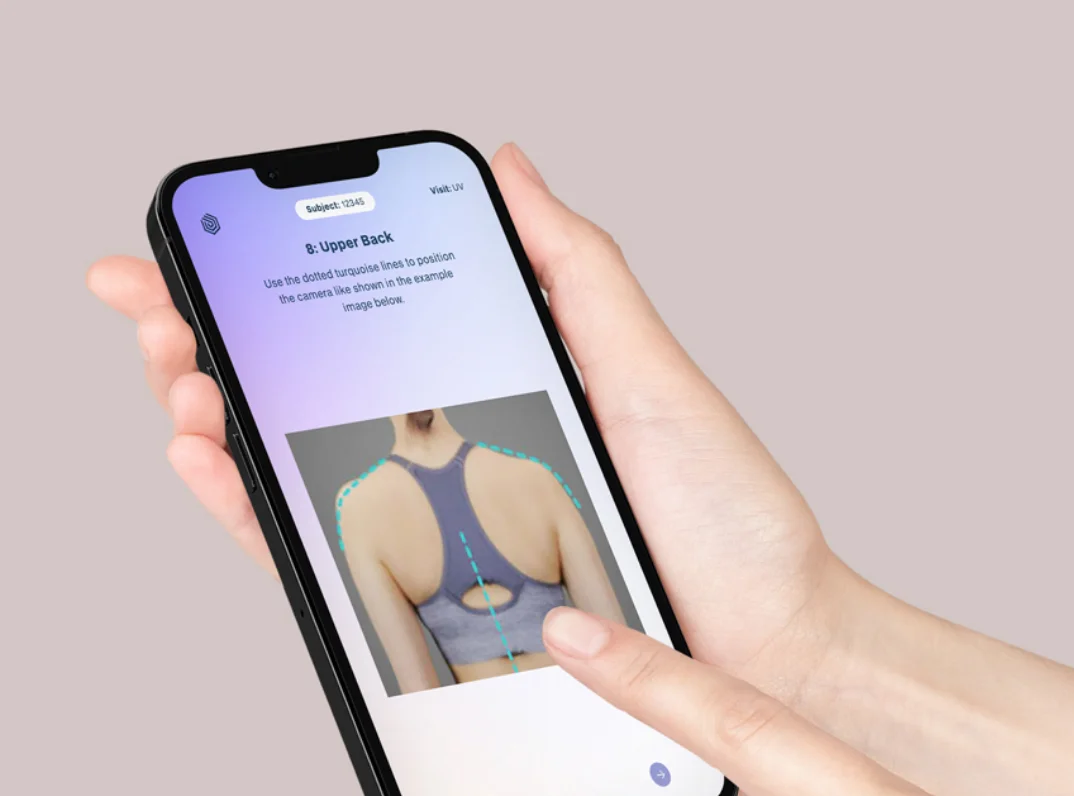

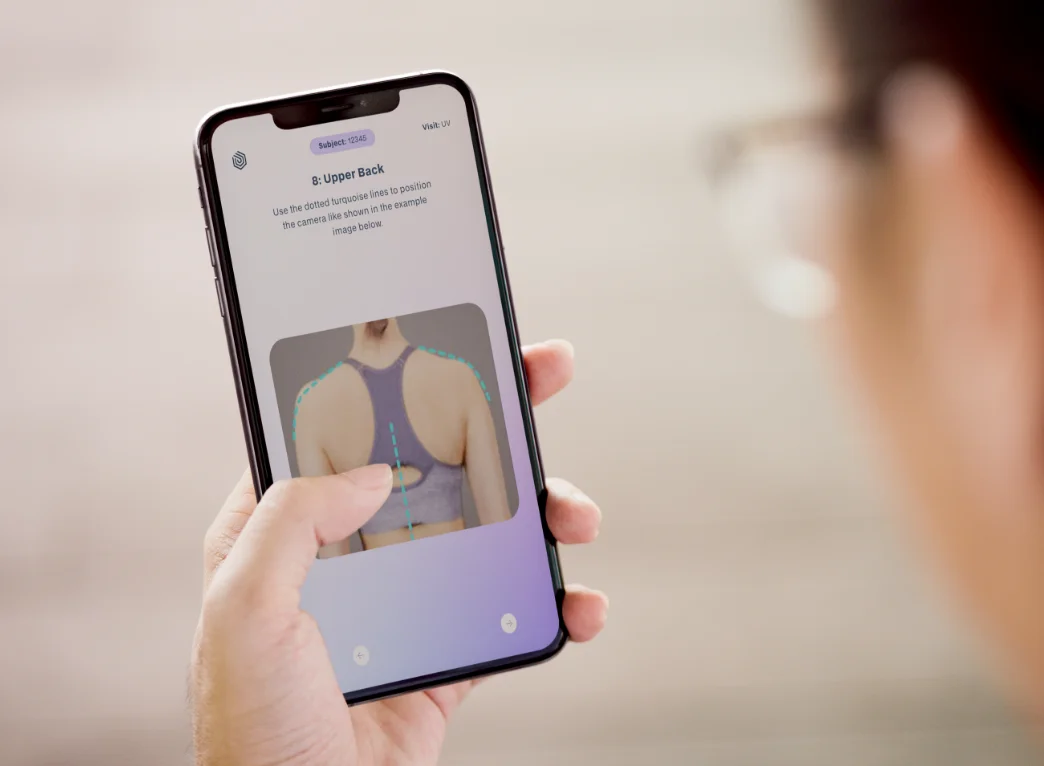


We are your partner for study specific solutions that exactly meet your needs
Artificial Intelligence
Advancing Possibilities with AI
At the heart of our company’s mission is a dedication to innovation, ensuring that advancements are pursued where they offer real value rather than for their own sake. This approach is particularly evident in our adoption of artificial intelligence (AI) across the organisation, which is guided by comprehensive risk-benefit analyses, regulatory compliance, and a strong commitment to scientific integrity. Above all, protecting our client data is our highest priority; we apply measures to ensure that client information remains private and secure at every step of our AI journey.
Our AI initiatives are purposefully designed to support, not replace, scientific and regulatory rigor. We are steadfast in our commitment to subject safety, data integrity, and compliance across every function we touch. We carefully select each AI tool to ensure it is appropriately suited to its intended purpose—much like choosing the correct instrument for a specific task, as one would not use a tractor to plant a tomato. Selecting the right AI solution is essential for effectiveness and precision.
Our AI initiatives are purposefully designed to support, not replace, scientific and regulatory rigor. We are steadfast in our commitment to subject safety, data integrity, and compliance across every function we touch. We carefully select each AI tool to ensure it is appropriately suited to its intended purpose—much like choosing the correct instrument for a specific task, as one would not use a tractor to plant a tomato. Selecting the right AI solution is essential for effectiveness and precision.
The AI Expert Group: Pioneering the Future
Our dedicated AI Expert Group stands at theforefront of this transformation. This cross-functional team actively scoutsfor emerging AI technologies, rigorously tests new functionalities, andcollaborates closely with our subject matter experts across departments. Byidentifying and evaluating cutting-edge AI use cases (see below), our experts ensure we remain ahead of the innovation curve.
With Humans-in-the-Loop (HITL) AI Enhances Research Value
- By integrating HITL principles, we ensure that artificial intelligence is not just thoughtfully applied but also guided by expert oversight. This collaborative approach combines the strengths of advanced AI technologies with the critical judgment, experience, and ethical standards of our research professionals. As a result, AI solutions enhance research value while maintaining scientific integrity, data privacy, and regulatory compliance—always keeping humans at the center of decision-making.
- Study Feasibility: AI-powered feasibility analysis streamlines clinical research by evaluating protocol requirements against local factors like standard care, regulations, and infrastructure. By aligning criteria with regional medical practices, AI supports identifying barriers and unique study features, while real-world data and predictive analytics improve site selection and patient recruitment. This comprehensive approach speeds up planning and supports informed, locally relevant decisions.
- End-to-End Data Stewardship: From defining key data elements to managing validation, integration, and transformation – AI ensures consistency, not complexity.
- Medical Oversight Support: AI assists in planning targeted medical review by highlighting safety signals and suggesting alternative review strategies—enabling efficient, scientifically sound data oversight while preserving expert-driven decisions.
- Medical Coding Automation: Our intelligent auto-encoding solution delivers accurate, consistent, and high-performing harmonisation of diverse datasets—especially in large-scale or multilingual studies—to standard MedDRA and WHODrug dictionaries.
- Medical Writing: Intelligent matching algorithms and GPT-based AIs (including agentic AI) support document creation, translation, and audience-specific adaptations.
- System Validation Optimisation: Implement intelligent test planning, scenario development, and comprehensive documentation to improve validation efficiency while maintaining full compliance.
- Case Narrative Drafting: Large Language models accelerate clinical writing workflows, supporting—but not replacing—scientific judgment. Predictive Intelligence Targeted forecasting tools provide scenario planning and operational insights when backed by high-quality data.
Partnering for Progress
We recognize that genuine innovation may be achieved through collaboration. Accordingly, we are partnering with leading AI providers to ensure our clients gain access to the most current advancements in artificial intelligence.
Looking Forward
By leveraging AI, we enable smarter, more agile operations, helping our clients achieve their goals with confidence. Together, we are shaping a future built on intelligence and collaboration.
CTMS
Comprehensive Clinical Trial Management System
SCOPE understands the need for flexibility depending on your specific project. We offer our custom full scale CTMS solution, INSIGHT, which is based on SharePoint and due to its integration with the Office Suite and our custom extensions, is particularly strong in document management and cooperation. Furthermore, we have a preferred CTMS vendor, BSI (BSI Business Systems Integration AG), who offers an integrated eTMF. Depending on your project set up and technical preferences, we will suggest the optimal CTMS solution to you or accommodate your preferred CTMS vendor.
Key CTMS Features:
- Template Management
- Document Creation, Review & Approval Workflows
- Electronic Signature & Signature Workflows
- eTMF Integration
- Project Tracking & Visualisation
- Protocol Deviation Management
- Data Import of subject recruitment data & programmatically detected protocol deviations
- Collaboration Tools
- Configuration & Customisation
- Project Planning
- Global Contact Database
- Technical Support
- Quality & Regulatory Compliance
eTMF
Electronic Trial Master File (eTMF)
At SCOPE, we are experienced with various eTMF providers including TransPerfect, BSI, Veeva, Medidata and Phlexglobal. Our preferred eTMF solutions are Trial Interactive from TransPerfect and BSI eTMF, as they meet regulatory requirements at a fair market value, however we are flexible to work with other eTMF solutions as well.
BSI eTMF, being integrated directly with BSI CTMS, provides the advantage an efficiency gains of a one-stop shop solution. Documents created in the CTMS show up in the eTMF right away together with all assigned metadata, making any further document sending or processing obsolete.
INSIGHT allows for an application programming interface (API) based integration with Trial Interactive eTMF. The partnership between TransPerfect and SCOPE is providing significant advantages to sponsors, including quicker set-up times for new trials, reduced initial costs, and more efficient file management. These improvements not only ensure the timely management of Trial Master Files but also optimize our budget for TMF maintenance, demonstrating our commitment to leveraging innovative solutions for more cost efficient and effective clinical trial management.
BSI eTMF, being integrated directly with BSI CTMS, provides the advantage an efficiency gains of a one-stop shop solution. Documents created in the CTMS show up in the eTMF right away together with all assigned metadata, making any further document sending or processing obsolete.
INSIGHT allows for an application programming interface (API) based integration with Trial Interactive eTMF. The partnership between TransPerfect and SCOPE is providing significant advantages to sponsors, including quicker set-up times for new trials, reduced initial costs, and more efficient file management. These improvements not only ensure the timely management of Trial Master Files but also optimize our budget for TMF maintenance, demonstrating our commitment to leveraging innovative solutions for more cost efficient and effective clinical trial management.
Key Features of our eTMF Solution:
- Optimised costs: Study-specific eTMFs are built from an existing master configuration which is continuously developing while eTMF technology advances. The master configuration provides minimal set-up costs for sponsor while including years of TMF experience.
- Quick set-up times: The existing TMF configuration enables TMF go-life within weeks while requiring minimal sponsor involvement.
- TMF Reference Model: The existing TMF configuration is based on the TMF Reference / Standard Model.
- Inspection Readiness: Tight integration between the eTMF and CTMS in combination with SCOPE’s TMF Plan ensures inspection readiness at all times. In addition to usual TMF completeness checks which compare against manually created events and placeholders, SCOPE’s TMF completeness is assessed against the entirety of all documents in the CTMS / INSIGHT, thereby providing a more relevant TMF expectation at reduced efforts and costs.
EDC / ePRO / RTMS
Electronic Data Capture (EDC) and Electronic Patient-Reported Outcomes (ePRO)
SCOPE works with a range of EDC, ePRO and RTSM (Randomization and Trial Supply Management) vendors to provide flexible and reliable data capture solutions. Our partnerships with vendors like Viedoc, Merative and Oracle allow us to builds study specific data collection solutions that are fit for purpose for each individual project. By combining deep system expertise with a pragmatic, patient centric mindset, we transform even complex trial designs into clear, intuitive and manageable processes, while maintaining full control, oversight and data integrity throughout the study lifecycle.
Key Features of our EDC, ePRO and RTSM Solutions:
- Study Specific System Design: Each system is purpose built for the study at hand. Data collection tools, workflows and logic are designed around protocol requirements, operational workflows, and the needs of sites and patients.
- Pragmatic, Patient Centric Data Capture: User friendly interfaces, intuitive designs, and streamlined visit flows reduce burden for sites and patients while enhancing data quality and completeness.
- Powerful Functionality with Full Oversight: Configurations provide strong oversight, traceability and control, while the flexibility of modern platforms allows complex study requirements to be implemented in a clear and transparent manner.
- Flexibility to Handle Complexity: Advanced system capabilities enable complex trial designs, multi source data and dynamic workflows to be translated into straightforward, robust and reliable processes.
- Customisable, Risk Based Solutions: Flexible configurations allow fast, precise system setup tailored to study specific risks, ensuring solutions that fully support a Risk Based Quality Management (RBQM) approach.
Customised Solutions
Customised Solutions
Regardless of the dimensions of your study, we can provide the technical solution that works for you. In the last years, we have successfully set up and used study specific solutions like a subject randomization system and a study specific IP allocation system.
Our most recent opus, a custom mobile app, was developed to document evidence in a study in the dermatological area with ease. The app was strongly tailored to the needs of the sponsor – in time and budget with proven quality. From concept, requirements management, risk assessment, implementation and validation to user support, platform maintenance, product improvement and data archival – SCOPE covers the full product lifecycle.
Our most recent opus, a custom mobile app, was developed to document evidence in a study in the dermatological area with ease. The app was strongly tailored to the needs of the sponsor – in time and budget with proven quality. From concept, requirements management, risk assessment, implementation and validation to user support, platform maintenance, product improvement and data archival – SCOPE covers the full product lifecycle.
eConsent
eConsent – Modernizing Informed Consent Through Trusted Partnerships
At SCOPE, we recognize that informed consent is a critical foundation of ethical clinical research. To enhance patient understanding, engagement, and regulatory compliance, we offer eConsent solutions through collaboration with leading technology vendors in the field. By partnering with specialised providers, we ensure that our clients benefit from state-of-the-art platforms that support multimedia content, electronic signatures, and real-time access—while maintaining full compliance with global regulatory standards.
Why We Use eConsent via Trusted Vendors:
- Enhanced Patient Comprehension through interactive and visual content
- Remote Access to consent materials on any device
- Streamlined Documentation with audit trails and version control
- Multilingual Capabilities for global trial deployment
- Regulatory Compliance with GDPR, 21 CFR Part 11, and local ethics requirements
- Seamless Integration with EDC, ePRO, and CTMS systems
Our vendor-based eConsent approach allows us to remain flexible and scalable — tailoring the solution to each study’s needs while ensuring a consistent, patient-centric experience.
Medical Coding
Medical Coding
SCOPE´s custom medical coding system is designed to support effortless and consistent coding of high volumes of clinical data. It enables data managers and medical monitors to efficiently code and review medical events and medications while maintaining full transparency, quality control, and regulatory alignment.
Key Features of our Medical Coding Solution:
- Advanced Autocoding with Enhanced Knowledge Base: Automated coding using MedDRA and WHO dictionaries, supported by an enhanced knowledge base built from validated historical coding decisions to improve accuracy, consistency and efficiency.
- Flexible Dictionary management: Full control over dictionary versions, updates and study specific configurations. materials on any device
- Efficient Code Assignment: Streamlined assignment of codes to medical terms, supporting both automatic and manual coding workflows.
- Medical Term Normalisation and Spelling Control: Consistent handling of spelling variants and term standardisation to support clean, analysis ready data.
- Integrated Query management: Support for queries related to medical coding, enabling clear communication and resolution workflows.
- Quality Control and Oversight: Built in quality checks and review processes ensure accuracy, traceability and inspection readiness of coded data.
Safety Database
Safety Database
Comprehensive Case Management
The safety database supports structured case booking and triage, comprehensive data entry, medical assessment, and narrative drafting. Sponsor assessments can be documented within the system, allowing the case evaluation to be reviewed and updated prior to finalisation.
Coding and Documentation
- Integration of current WHO Drug Dictionary and MedDRA for accurate and consistent coding
- Case file composition and narrative creation directly within the system
- Documentation of medical and sponsor assessments within the case record
Data Integrity, Qualityand Control
- Duplicate case detection to prevent multiple processing of the same case and to maintain a single, consistent case record
- Version control of cases, maintaining full transparency of changes
- Case locking to prevent unauthorised modifications following finalisation
- Comprehensive audit trail capturing all user actions and data changes
- Integrated quality control functionality supporting internal review and approval workflows
Security and User Management
- Role based user access control, ensuring appropriate permissions and segregation of duties
- Secure handling of sensitive safety data in line with GxP principles and data protection requirements
Oversight, Transparency and Reporting Support
- User Interface (UI) controls for oversight, providing real time visibility into case status and timelines
- Case search and sorting capabilities for efficient retrieval and review of safety data case and line listing exports to support oversight, reconciliation and reporting activities
Safety Regulatory Reporting
Safety Regulatory Reporting
The SCOPE Expedited and Periodic Reporting Information System (SCORIS) is a dedicated module for regulatory safety reporting, designed to manage expedited and periodic reporting obligations and to provide oversight of reporting compliance to institutions and investigators within clinical studies.
Regulatory Requirements & Reporting Setup
- Central database of regional and country specific safety reporting requirements
- Identification of applicable reporting obligations per case or reporting activity
Safety Report Distribution
- Automatic generation of distribution lists for reportable cases and periodic safety reports, leading regulatory documents and submission of safety reports to the relevant institutions and investigator
Submission Tracking & Compliance Oversight
- Identification of approaching regulatory reporting deadlines
- Tracking of submission dates and submission status
- Built in tool for safety reporting compliance calculation and performance monitoring
Download now!
Fact Sheet
DownloadContact
Let`s grow together!
SCOPE International AG
Konrad-Zuse-Ring 18
68163 Mannheim, Germany
+49 621 429-390
contact@scope-international.com
contact usKonrad-Zuse-Ring 18
68163 Mannheim, Germany
+49 621 429-390
contact@scope-international.com

